What's involved in taking part?
We will ask your medical team to send us a sample of your cancer taken either from the tumour removed during surgery or biopsy before surgery.
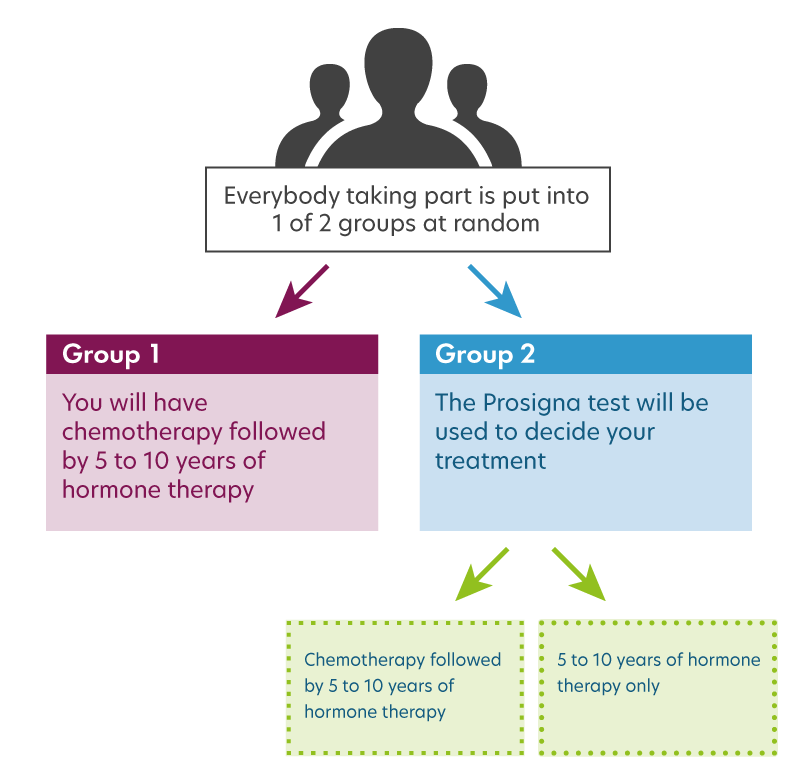
OPTIMA is a randomised controlled trial. A randomised controlled trial offers the best way to compare the effectiveness of Prosigna with the current standard approach for deciding when to give a patient chemotherapy.
We use a process call randomisation to put patients into one of two groups – Group 1 and Group 2. Patients have an equal chance of being in either group.
Neither you nor your doctor can decide which group you are in. We do this to avoid influencing the results in any way. We want to have two groups of patients that are as similar as possible in all ways except for the way in which their treatment is decided. This will enable us to make a fair comparison of chemotherapy decisions using Prosigna with standard practice.
What treatment might I receive?
The Group that you are in will determine your ongoing treatment.
- Group 1 you will receive chemotherapy followed by hormone therapy – the standard treatment that you would be offered outside the study. The Prosigna test will not decide your treatment.
- Group 2 the Prosigna test will decide your treatment. Depending on the result of the test, you will either have chemotherapy followed by hormone therapy or have hormone therapy without chemotherapy.
Will I know what treatment group I am in?
If you go straight to hormone therapy you will know that you are in Group 2. Prosigna was used on a sample of your cancer and the result was that you are unlikely to benefit from chemotherapy.
If you have chemotherapy followed by hormone therapy, you and your Dr will not know if you are in Group 1, and so are having chemotherapy as standard or you are in Group 2, where your tissue sample was tested and the result was that you would likely benefit from chemotherapy.
How long will it be before I know what treatment I will have?
It usually takes between two to three weeks to receive news of the treatment you will receive. We know that this is a safe time to wait.
Quality of life
To help us understand both the financial and personal impact of your treatment, we will ask you to fill out a health-related quality of life and economic questionnaires when you agree to join the study and then at:
- 3 months
- 6 months
- 12 months
- 24 months
Qualitative Recruitment Study
We want to improve how information about OPTIMA and other clinical studies is provided to patients. To help with this, we are inviting patients to take part in the OPTMA Qualitative Recruitment Study (QRS). The QRS has two elements, audio-recording consultations and talking with you about your decision.

We want to learn more about how patients are invited to take part in OPTIMA. To explore this, your oncologist or research nurse may ask if you’d be happy to have your oncology appointment audio recorded. We’ll use the recordings to gain an insight into how your medical team explained the study to you.

We also want to understand what patients take into account when deciding about OPTIMA. To gain an insight, we would like to talk with some patients. Your research nurse may ask if you would be happy to have a conversation with a researcher.
You can take part in the OPTIMA Qualitative Recruitment Study regardless of whether or not you agree to join the OPTIMA study.
The QuinteT team at the University of Bristol are running the Qualitative Recruitment Study.

